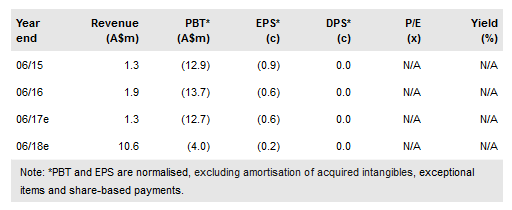
Prima Biomed Ltd (AX:PRR) has presented encouraging early signs of efficacy from the TACTI-mel trial of IMP321 in combination with Keytruda, with one of the six melanoma patients in the first (1mg/kg) cohort experiencing a complete response. Recruitment in the second cohort is complete and the final cohort is expected to be fully recruited by Q317. Preliminary efficacy data from the 15-patient, run-in phase of the AIPAC breast cancer study are expected mid-year (recruitment in the 226-patient Phase IIb component is ongoing). Our valuation is unchanged at A$252m (12c per share).
A complete response in low-dose TACTI-mel cohort
The TACTI-mel trial is using IMP321 (Prima’s LAG-3-based antigen presenting cell activator) to enhance efficacy in melanoma patients who have had a suboptimal initial response to the PD1 immune checkpoint inhibitor Keytruda. A presentation to the Immune Checkpoint Inhibitors conference in Boston showed that that one of the six patients (17%) in the first cohort experienced a complete response (CR). Even though there is only one patient with a CR, the initial CR rate compares favourably with rates of 2-6% seen in Merck’s Phase III trials of Keytruda monotherapy in melanoma. The combination has been well tolerated so far. Recruitment in the third and highest (30mg) dose cohort is expected to complete in Q317, so we expect efficacy data from the final cohort in H118.
To read the entire report Please click on the pdf File Below